|
6) Under which set of conditions is a chemical reaction most likely to be spontaneous. Okay, so now we can move to be so here we see we have s to gas and s eighth solid. 2) Which has the greatest absolute entropy (A) one mol of C(s) at 25 C. Since there is no disorder in this state, the entropy can be defined as zero. Imagine cooling the substance to absolute zero and forming a perfect crystal (no holes, all the atoms in their exact place in the crystal lattice). 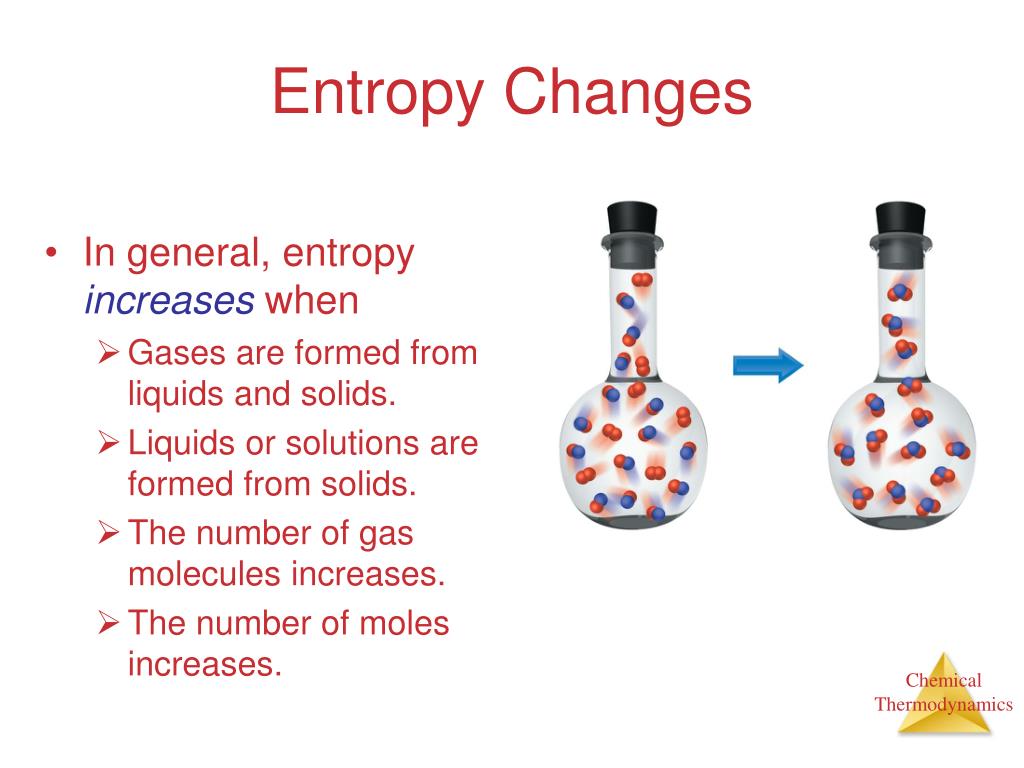
And so we see that we have s two and s eight and therefore s eight is going to have the greatest amount of entropy because it is the largest molecule. The absolute entropy of any substance can be calculated using equation (1) in the following way. So the next thing we can ask is which is larger because the larger ba molecule, the more confirmations it can adopt in the more ways that it can move around. So then we can ask what state of matter? Because we know that gases have the highest entropy, and if something are solid that it would have very low interview. And we also know that entropy depends on molar mass so the molar mass of chlorine is higher than fluorine so the absolute entropy per mole of chlorine is. So we can compare the two and we see that we have one mole of each and therefore the same number of particles. We know the entropy of Gaseous state is higher than liquid and solid. Since entropy increases logarithmically with the number of microstates, the most probable distribution is therefore the one of greatest entropy. (b) Calculate the enthalpy of vaporization of methanol, assuming that rS doesnt depend on temperature and taking the boiling point of methanol to be 64.6C. (a) Calculate the entropy change for the vaporization of 1 mol methanol (use data from Table 16.1 or Appendix J). In this case, S fus (6.01 kJ/mol)/ (273 K) 22.0 J/ (molK) S sys. The standard molar entropy of methanol vapor, CH3OH(g), is 239.8 J K1 mol-1. 6: Thermograms Showing That Heat Is Absorbed from the Surroundings When Ice Melts at 0C. So when we're trying to decide which has the greatest entropy, the first consideration is which has the greatest number of moles and therefore particles, because we know that the higher the number of particles that we have, the more entropy. The amount of heat lost by the surroundings is the same as the amount gained by the ice, so the entropy of the universe does not change. 
For this question were asked to compare four different sets of molecules and to determine which of the two in Egypt set has the greatest entropy.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
